Abstract:
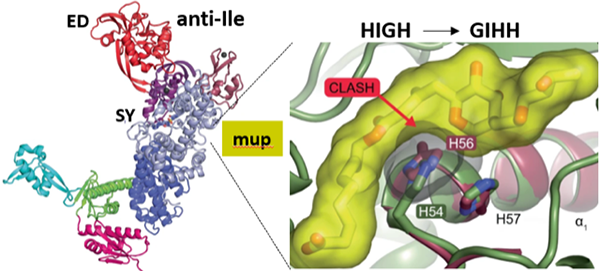
Aminoacyl-tRNA synthetases (AARSs) decode the genetic information by linking the cognate pairs of amino acids and tRNAs. In doing so, AARSs face a challenge in discriminating against non-cognate amino acids that are highly similar to the cognate ones. These near-cognate amino acids are often mischarged to the tRNA in the synthetic active site and eliminated at the distant editing domain to keep translation errors within a tolerable limit. Isoleucyl-tRNA synthetase (IleRS) is editing class I AARS responsible for introducing isoleucine into proteins. My lab has been working on the mechanisms of the IleRS synthetic and editing reactions for years. Recently, we found that non-proteinogenic amino acid norvaline may jeopardize the accuracy of Ile-tRNAIle synthesis, making the IleRS editing domain a fortress of canonical translation. We further discovered that opposing to the prevailing view, the editing domain may also clear non-cognate amino acids that do not pose accuracy threats. The observed broad selectivity is compensated by the exquisite specificity in preventing hydrolysis of Ile-tRNAIle, the latter ensured by negative catalysis. In bacteria, two IleRS types (1 and 2) may perform housekeeping roles. We questioned why IleRS2 enzymes, which are resistant to the natural antibiotic mupirocin, did not displace IleRS1 known to be efficiently inhibited by mupirocin binding to the synthetic site. Detailed kinetic analysis revealed that IleRS1 enzymes support faster translation than IleRS2 and thus are essential for faster-growing bacteria. The real surprise, however, came from discovering that IleRS2 may harbor a natural variation of the class I AARS signature motif, the HIGH motif. The altered HIGH motif, which is not found or tolerated in IleRS1, brings mupirocin hyper-resistance without compromising IleRS2 catalysis. The structural basis of the altered motif accommodation and its link to hyper-resistance will be discussed.
Speaker: Dr. Ita Gruic-Sovulj, University of Zagreb Faculty of Science, Zagreb, Croatia.
Host: Liam Longo, ELSI.
Date: Wed, 20 Sep., 16:00-17:00 JST
Venue: Online
