[This is a joint press release with Georgia Institute of Technology]
Life’s first alphabet was likely small, but surprisingly powerful
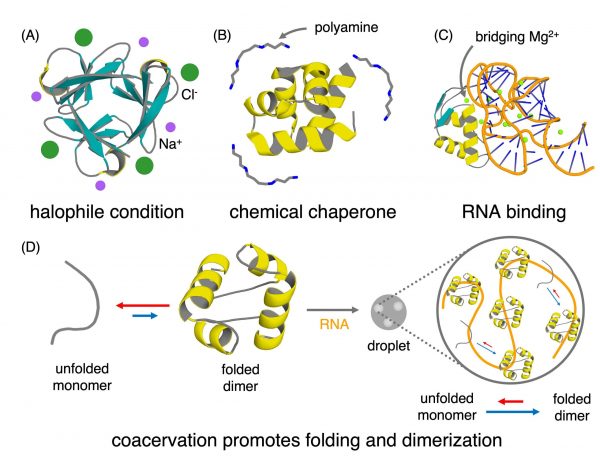
The first proteins may have emerged in chemically rich environments. These environments contain molecules like salts that stabilize protein structures, making early folding easier. Credit: Koh Seya, Alfie-Louise Brownless, Shina Caroline Lynn Kamerlin, and Liam M. Longo
How did the earliest life on Earth build complex biological machinery with so few tools? A new study explores how the simplest building blocks of proteins, once limited to just half of today’s amino acids, could still form the sophisticated structures life depends on.
The paper, “The Borderlands of Foldability: Lessons from Simplified Proteins,” is a meta-analysis of six decades of protein research and reveals that ancient proteins may have been far more complex and dynamic than previously thought.
Recently published in Trends in Chemistry, the study includes researchers from the Earth-Life Science Institute (ELSI) at the Institute of Science Tokyo, including Specially Appointed Associate Professor Liam M. Longo and Graduate Student Koh Seya, alongside Georgia Tech researchers Lynn Kamerlin, Professor in the School of Chemistry and Biochemistry and Georgia Research Alliance Vasser Woolley Chair in Molecular Design, and Quantitative Biosciences Ph.D. candidate Alfie‑Louise Brownless.
The research has implications ranging from the origins of life and the search for life in the universe to modern approaches in protein design and biotechnology.
If proteins are the scaffolding of life, amino acids are the components that make up that scaffolding. Today, an average protein is constructed from a chain of about 300 amino acids, involving 20 different types of amino acids. Proteins fold when these chains twist into a specific three-dimensional shape, creating structures critical for biology.
However, while these folds are essential, exactly how a protein arrives at its final structure remains a major scientific question. Randomly sampling all possible configurations would take longer than the age of the universe, yet proteins reliably fold in a fraction of a second.
This long-standing puzzle, often referred to as Levinthal’s paradox, highlights a fundamental mystery. Proteins fold rapidly into precise structures, but the underlying rules that guide this process are still not fully understood.
According to Koh Seya, a member of the Longo Group and first author on this work, “Early proteins likely had access to only a limited set of amino acid types, perhaps just 10 to 12. That’s about half of the current repertoire. Would building a protein using just a few amino acid types be like writing a story using only the letters ‘A’ through ‘L’? Would it severely restrict the possibilities?”
The study shows that this is not necessarily the case. Even with a restricted “alphabet”, proteins can form complex folds. By examining simplified proteins, researchers found that a combination of sequence design and environmental conditions can support folding.
Certain environments, such as those with high salt concentrations, may have helped stabilise early proteins. In addition, simple peptides could associate with one another, forming larger and more complex structures through cooperative interactions.
The findings suggest that early proteins were not necessarily simple in behaviour, even if their building blocks were limited. Instead, they may have relied on interactions, assembly, and environmental support to achieve functional complexity.
“This work helps us understand how complex protein structures could emerge from very simple starting points,” said Liam M. Longo. “If early proteins were able to fold and function with a limited set of building blocks, it suggests that these structures are remarkably robust, and that their emergence early in life’s history was not a matter of luck, but perhaps an inevitability.”
By exploring how proteins can form under constrained conditions, it becomes possible to better understand both the origins of life and how to design new proteins today. These insights connect fundamental questions about early Earth with modern challenges in biotechnology and medicine.
The team is continuing to explore environments that could mimic conditions on early Earth, aiming to better understand how such systems may have supported the emergence of protein structure and function.
The study also highlights broader questions about how proteins evolved, how different folds are connected, and how simple molecular systems transitioned into the complex biological machinery seen today.
“Understanding these processes may not only shed light on the origins of life on Earth, but also guide the search for life beyond our planet and inform the design of new proteins for medical and technological applications,” said Lynn Kamerlin.
Reference
Koh Seya (瀬谷巧) 1, Alfie-Louise R. Brownless 2, Shina C.L. Kamerlin 2 3 4, Liam M. Longo 5 6, The borderlands of foldability: lessons from simplified proteins, Trends in Chemistry, DOI: 10.1016/j.trechm.2026.03.001
- Department of Life Science and Technology, Institute of Science Tokyo, Tokyo 152-8550, Japan
- School of Chemistry and Biochemistry, Georgia Institute of Technology, Atlanta, GA 30332, USA
- Department of Chemistry, Lund University, Lund 22011, Sweden
- School of Chemical and Biomolecular Engineering, Georgia Institute of Technology, Atlanta, GA 30332, USA
- Earth-Life Science Institute, Institute of Science Tokyo, Tokyo 152-8550, Japan
- Blue Marble Space Institute of Science, Seattle, WA 98104, USA
*Corresponding author’s email: llongo@elsi.jp
Contacts:
Thilina Heenatigala
Director of Communications
Earth-Life Science Institute (ELSI),
Institute of Science Tokyo
E-mail: thilinah@elsi.jp
Tel: +81-3-5734-3163 / Fax: +81-3-5734-3416
Liam M. Longo
Specially Appointed Associate Professor
Earth-Life Science Institute (ELSI),
Institute of Science Tokyo
E-mail: llongo@elsi.jp
