[Press Release]
Researchers show that enzymes with enhanced enzyme diffusion can function as Maxwell’s demon by utilising information from past reactions to actively shift away from equilibrium and control the directionality of chemical reactions.
Figure 1. Enzymes as Maxwell’s Demon
An artistic illustration depicting “Maxwell’s demon” modulating the diffusion speed of an enzyme. The demon represents the physical mechanism by which the enzyme uses information to break chemical equilibrium. Credit: Tetsuhiro S. Hatakeyama, ELSI
Living cells are sustained by countless chemical reactions that must be carefully regulated to maintain internal order and function. Enzymes play a central role in this process, accelerating reactions that would otherwise proceed too slowly to support life. Traditionally, enzymes have been viewed as passive catalysts—speeding up chemical reactions without influencing their final balance. However, how enzymes might contribute to the regulation of chemical states beyond simple catalysis remains an open question in biology.
A study led by researchers from Earth-Life Science Institute (ELSI) at Institute of Science Tokyo, explain the biological consequences of enhanced enzyme diffusion or mobility that is observed during catalysis. Their new study proposes that enzymes behave similar to the theoretical “Maxwell’s demon” and can utilise information from past reactions to actively shift subsequent reactions away from equilibrium. This is a fundamentally new role for enzymes with implications in fine metabolic regulation.
It all began with the observation of the phenomenon of enhanced enzyme diffusion (EED), in which enzymes transiently move faster after catalysis. Instead of treating enhanced diffusion as a secondary effect, the researchers asked whether it could play an active functional role in chemical reactions. Their work provides a fundamental explanation for how EED could influence reaction dynamics at the macroscopic level.
In the study, the researchers simulated the scenario where chemical energy generated during a catalytic reaction is utilised by the enzymes to transiently increase mobility. They tested whether this change in motion altered subsequent reactions; in particular, they studied the composition of substrates and products. In their simulation analysis, they observed that the ratio of substrate to product exhibited a clear deviation from the expected chemical equilibrium.
How do simple diffusion changes break the chemical equilibrium? “We struggled to understand the physical mechanism driving this shift,” says Associate Professor Tetsuhiro S. Hatakeyama, co-author of the study. “The most challenging aspect was bridging the gap between the simulation results and the theoretical understanding.”
The key insight came from recognising that the enzyme’s behaviour resembled a famous thought experiment known as Maxwell’s demon, which describes an imaginary being that uses information about molecular motion to create order without doing work, seemingly violating the second law of thermodynamics. In this study, the enzyme plays a comparable role through physical processes rather than intention. “Realising the analogy to ‘Maxwell’s demon’ was the critical conceptual leap that allowed us to view the enzyme as an agent performing measurement and feedback. This allowed us to understand that the biological behaviour of increased diffusion is a form of ‘memory’ and connected to information thermodynamics,” explains co-author Kunihiko Kaneko, Visiting Researcher of ELSI.
Based on this, the researchers constructed a theoretical model where the transient increase in motility served as a “memory” of the enzyme’s immediate past reaction event. The enzyme used this information to leave the product molecules, thereby eliminating the probability of the reverse reaction. This behaviour disrupts the delicate balance between forward and reverse reactions and drives the system to a new steady state that deviates from the chemical equilibrium. Adding to the significance of this study is the finding that this phenomenon is not limited to the theoretical world but is biologically possible within the parameters of actual enzymes, such as urease.
One of the most significant implications of this finding is the possibility that EED may have existed in primitive “proto-enzymes” that may have utilised the heat or energy from chemical reactions to drive non-equilibrium reactions purely through physical motility changes. EED may be the potential “missing link” in prebiotic chemistry, serving as a simple, physical principle that paved the way for the emergence of life.
Overall, this study overturns the traditional passive role of enzymes by showing that enzymes can process information to actively control the directionality of chemical reactions. It also provides a concrete, biological realisation of the theoretical “Maxwell’s demon” and suggests that nature may have been utilising information-to-energy conversion mechanisms in biomolecules all along. Most notably, this study clears the mystery surrounding the biological significance of EED.
Looking ahead, further investigation will be needed to understand how this mechanism operates within living cells. Sharing their vision for this study, Hatakeyama says, “Theoretically, we aim to explore how this ‘demon-like’ behaviour affects larger metabolic networks. Does the cell use this mechanism to regulate metabolic flux or create spatial organisation?”
Together, these findings add a new dimension to biochemical regulation and could reshape our understanding of how enzymes function within living systems.
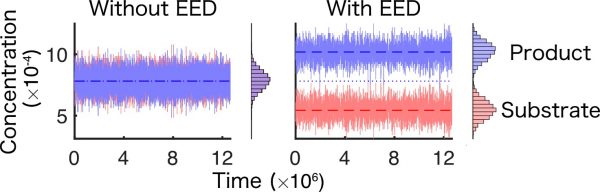
Figure 2. Deviation from chemical equilibrium by enhanced enzyme diffusion]
Comparison of molecular concentrations with and without enhanced enzyme diffusion (EED). In the absence of EED, the concentrations of the product and substrate align with chemical equilibrium (set here to be equal). However, in the presence of EED, the balance between forward and reverse reactions is broken, resulting in a steady-state increase in product concentration. Credit: Hatakeyama et al. Adapted from Physical Review Letters
Figure 3. Deviation from chemical equilibrium by enhanced enzyme diffusion
Comparison of molecular concentrations with and without enhanced enzyme diffusion (EED). In the absence of EED, the concentrations of the product and substrate align with chemical equilibrium (set here to be equal). However, in the presence of EED, the balance between forward and reverse reactions is broken, resulting in a steady-state increase in product concentration. Credit: Hatakeyama et al. Adapted from Physical Review Letters
Reference
Shunsuke Ichii1, 2, Tetsuhiro S. Hatakeyama3*, and Kunihiko Kaneko3, 4, Enzyme as Maxwell’s Demon: Steady-State Deviation from Chemical Equilibrium by Enhanced Enzyme Diffusion, Physical Review Letters, DOI: 10.1103/flv6-zw1v
- Department of Physics, The University of Tokyo, 7-3-1 Hongo, Bunkyo-ku, Tokyo 113-0033, Japan
- Center for Biosystem Dynamics Research, RIKEN, 6-2-3 Furuedai, Suita, Osaka 565-0874, Japan
- Earth-Life Science Institute, Institute of Science Tokyo, 2-12-1 Ookayama, Meguro-ku, Tokyo 152-8550, Japan
- Niels Bohr Institute, University of Copenhagen, Blegdamsvej 17, 2100 Copenhagen, Denmark
*Corresponding author’s email: hatakeyama@elsi.jp
Contacts:
Thilina Heenatigala
Director of Communications
Earth-Life Science Institute (ELSI),
Institute of Science Tokyo
E-mail: thilinah@elsi.jp
Tel: +81-3-5734-3163 / Fax: +81-3-5734-3416
Tetsuhiro S. Hatakeyama
Associate Professor
Earth-Life Science Institute (ELSI),
Institute of Science Tokyo
E-mail: hatakeyama@elsi.jp
